A possible procedure would be to use a graduated cylinder and count the number of drops required to fill the cylinder to the 1, 2, 3 etc milliliter marks. Divide the participants into small working groups of 3 or 4 and have them brainstorm to answer the question. Then ask, "So if we know the number of drops, how can we determine the volume measured in milliliters of a number of drops?" This question should incite a number of responses. So how can we use these micropipets to measure volumes of liquids? Accept all answers and choose, "by counting the number of drops."Īssessment: Monitor the participation of the participants to make sure that all understand before moving along. We will be asked to add 1 milliliter of this or 5 milliliters of that so we need to know how much to add from the micropipette. Introduce this investigation by posing the following problem to the participants: Throughout the year you will be using micropipets filled with various liquids to carry out microscale experiments. Participants find the mass to volume ratio of oil.Participants find the mass to volume ratio of water.Participants graph mass and volume of water.Using the graph from the previous step, participants determine the mass of one drop of water.Participants graph the data from the previous step.Participants determine experimentally the mass of 5, 10, 15, 20, etc.Using the graph, participants find the volume of one drop of water.Participants graph the data obtained from the previous step.Participants determine experimentally the number of drops of water in whole numbered milliliters of water.Participants are introduced to the micropipet as a means to measure volume.Overview: Use the following as a guide to read through the procedures in their entirety before attempting to conduct this lesson: Graph paper can be printed free online (google search). If available, but not necessary - buret and volumetric flask. Water, cooking oil, electronic balances, 10 mL graduated cylinders, micropipets, and uncooked vermicelli or other pasta. 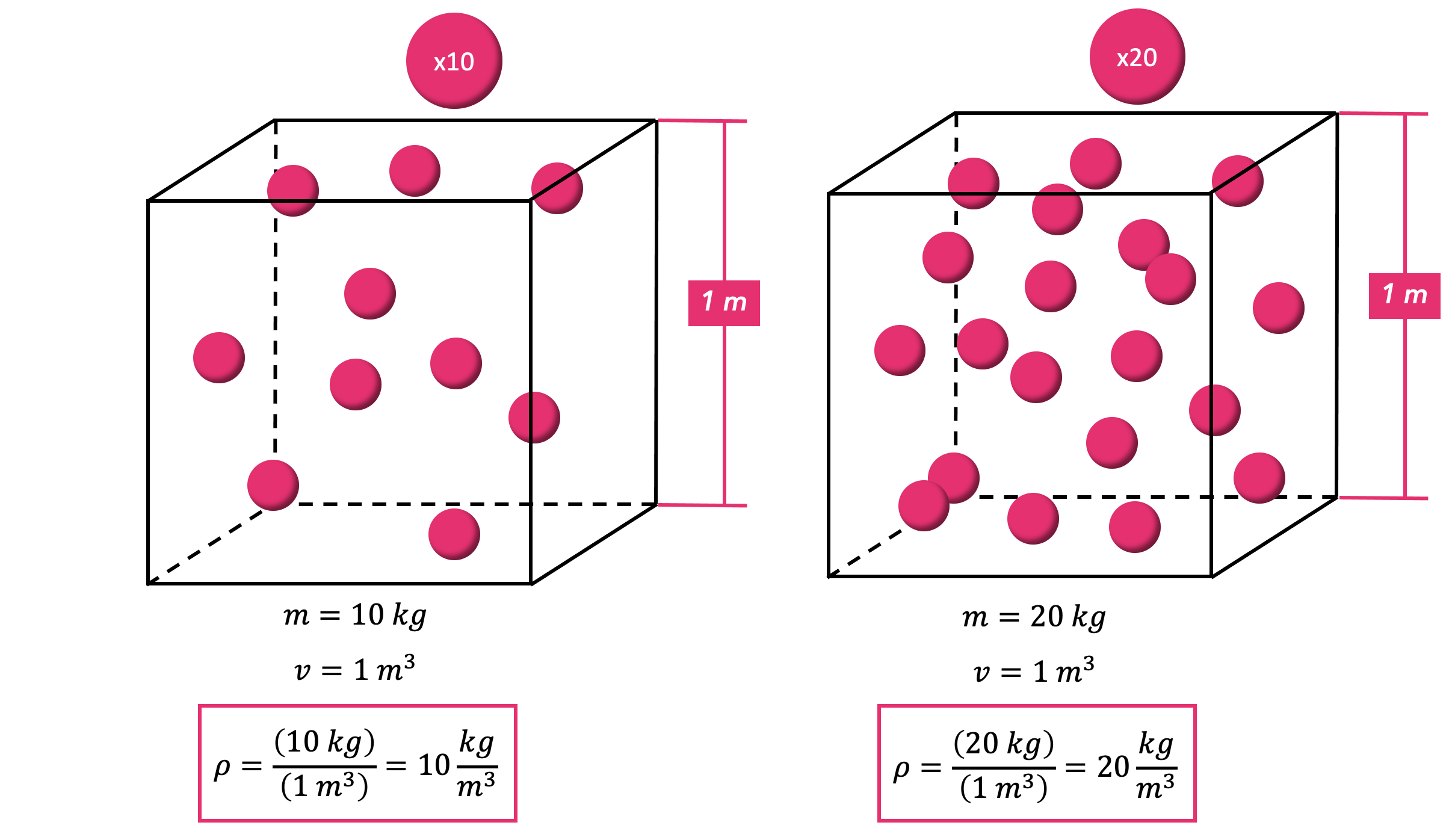
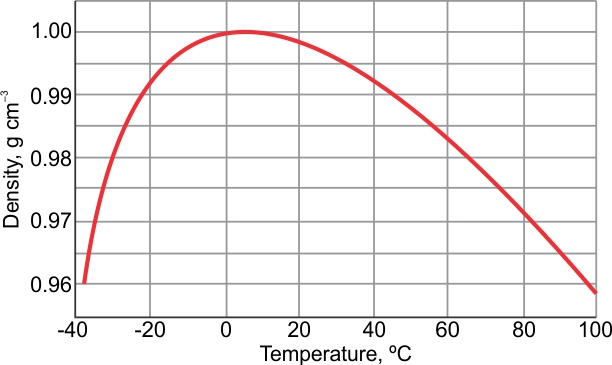
Participants will be able to produce and implement a lesson plan to lead their students through a discussion of how to graph and solve a problem using the graph.Participants will be able to graph the data.Participants will be able to gather data (mass, volume, and number of drops of water).Participants will be led through an exercise to devise a strategy to teach students how to properly graph data gained from massing and measuring the volume of different numbers of drops of water and finding the density of water and oil. Printer Friendly Version Determining the Density of Water - A Graphing Exercise = g/cc = g/ml = g/mL - they are all the same.Return to the Professional Development Module Index This structure consists of two hydrogen atomsĭivided by 16.02 = lbs/cu.ft. Maximum density does not occur when solidified. Fresh water has a maximumĭensity at around 4° Celsius. It expands rapidly adding about 9 % by volume. 
Substance on Earth that exists in all three physical states of matter: The density of water varies with temperature and impurities. % of the Earth's surface is covered with about 1.36 billion cubic kilometersĭensity of pure water is a constant at a particular temperature, andĭoes not depend on the size of the sample. Most animals and plants contain more than 60 % water by volume. Has a density (weight or mass) of about 1 g/cu.cm, 1 g/ml,ġ kg/liter, 1000 kg/cu.m, 1 ton/cu.m or 62.4 lb/cu.ft See other water links at the bottom of this informational page. We strongly implore you to conserve our natural resources, all of the, but especially our water. In the information below, you will find some interesting general data about water. 
Without water, there is no life on our Earth. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
